Definition
A nucleic acid is a chain of nucleotides which stores genetic information in biological systems. It creates DNA and RNA, which store the information needed by cells to create proteins. This information is stored in multiple sets of three nucleotides, known as codons.
How Nucleic Acids Work
The name comes from the fact that these molecules are acids – that is, they are good at donating protons and accepting electron pairs in chemical reactions – and the fact that they were first discovered in the nuclei of our cells.
Typically, a nucleic acid is a large molecule made up of a string, or “polymer,” of units called “nucleotides.” All life on Earth uses nucleic acids as their medium for recording hereditary information – that is nucleic acids are the hard drives containing the essential blueprint or “source code” for making cells.
For many years, scientists wondered how living things “knew” how to produce all the complex materials they need to grow and survive, and how they passed their traits down to their offspring.
Scientists eventually found the answer in the form of DNA – deoxyribonucleic acid – a molecule located in the nucleus of cells, which was passed down from parent cells to “daughter” cells.
When the DNA was damaged or passed on incorrectly, the scientists found that cells did not work properly. Damage to DNA would cause cells and organisms to develop incorrectly, or be so badly damaged that they simply died.
Later experiments revealed that another type of nucleic acid – RNA, or ribonucleic acid – acted as a “messenger” that could carry copies of the instructions found in DNA. Ribonucleic acid was also used to pass down instructions from generation to generation by some viruses.
Function of Nucleic Acids
Nucleic Acids Store Information Like Computer Code
By far the most important function of nucleic acids for living things is their role as carriers of information.
Because nucleic acids can be created with four “bases,” and because “base pairing rules” allow information to be “copied” by using one strand of nucleic acids as a template to create another, these molecules are able to both contain and copy information.
To understand this process, it may be useful to compare the DNA code to the binary code used by computers. The two codes are very different in their specifics, but the principle is the same. Just as your computer can create entire virtual realities simply by reading strings of 1s and 0s, cells can create entire living organisms by reading strings of the four DNA base pairs.
As you might imagine, without binary code, you’d have no computer and no computer programs. In just the same way, living organisms need intact copies of their DNA “source code” to function.
The parallels between the genetic code and binary code have even led some scientists to propose the creation of “genetic computers,” which might be able to store information much more efficiently than silicon-based hard drives. However as our ability to record information on silicon has advanced, little attention has been given to research into “genetic computers.”
Protecting the Information
Because the DNA source code is just as vital to a cell as your operating system is to your computer, DNA must be protected from potential damage. To transport DNA’s instructions to other parts of the cell, copies of its information are made using another type of nucleic acid – RNA.
It’s these RNA copies of genetic information which are sent out of the nucleus and around the cell to be used as instructions by cellular machinery.
Cells also use nucleic acids for other purposes. Ribosomes – the cellular machines that make protein – and some enzymes are made out of RNA.
DNA uses RNA as a sort of protective mechanism, separating the DNA from the chaotic environment of the cytoplasm. Within the nucleus, the DNA is protected. Outside of the nucleus, movements of organelles, vesicles, and other cellular components could easily damage the long, complex DNA strands.
The fact that RNA can act both as hereditary material and an enzyme strengthens the case for the idea that the very first life might have been a self-replicating, self-catalyzing RNA molecule.
Examples of Nucleic Acids
The most common nucleic acids in nature are DNA and RNA. These molecules form the foundation for the majority of life on Earth, and they store the information necessary to create proteins which in turn complete the functions necessary for cells to survive and reproduce. However, DNA and RNA are not the only nucleic acids. However, artificial nucleic acids have also been created. These molecules function in the same way as natural nucleic acids, but they can serve a similar function. In fact, scientists are using these molecules to build the basis of an “artificial life form”, which could maintain the artificial nucleic acid and extract information from it to build new proteins and survive.
Generally speaking, nucleic acids themselves differ in every organism based on the sequence of nucleotides within the nucleic acid. This sequence is “read” by cellular machinery to connect amino acids in the correct sequence, building complex protein molecules with specific functions.
Nucleic Acids and Genetics
The Genetic Code
Today, scientists know that the source code for cells is quite literally written in nucleic acids. Genetic engineering changes organisms’ traits by adding, removing, or rewriting parts of their DNA – and subsequently changing what “parts” the cells produce.
A sufficiently skilled genetic “programmer” can create the instructions for a living cell from scratch using the nucleic acid code. Scientists did exactly that in 2010, using an artificial DNA synthesizer to “write” a genome from scratch using bits of source code taken from other cells.
All living cells on Earth “read” and “write” their source codes in almost exactly the same “language” using nucleic acids. Sets of three nucleotides, called codons, can code for any given amino acid, or for the stop or start of protein production.
Other properties of nucleic acids may influence DNA expression in more subtle ways, such as by sticking together and making it harder for transcription enzymes to access the code they store.
The fact that all living cells on Earth “speak” almost the same genetic “language” supports the idea of a universal common ancestor – that is, the idea that all life on Earth today started with a single primordial cell whose descendants evolved to give rise to all modern living species.
From a chemical perspective, the nucleotides that are strung together to create nucleic acids consist of a five-carbon sugar, a phosphate group, and a nitrogen-containing base. The image below shows structural drawings of the four DNA and the four RNA nitrogenous bases used by living things on Earth in their nucleic acids.
It also shows how the sugar-phosphate “backbones” bond at an angle that creates a helix – or a double helix in the case of DNA – when multiple nucleic acids are strung together into a single molecule:
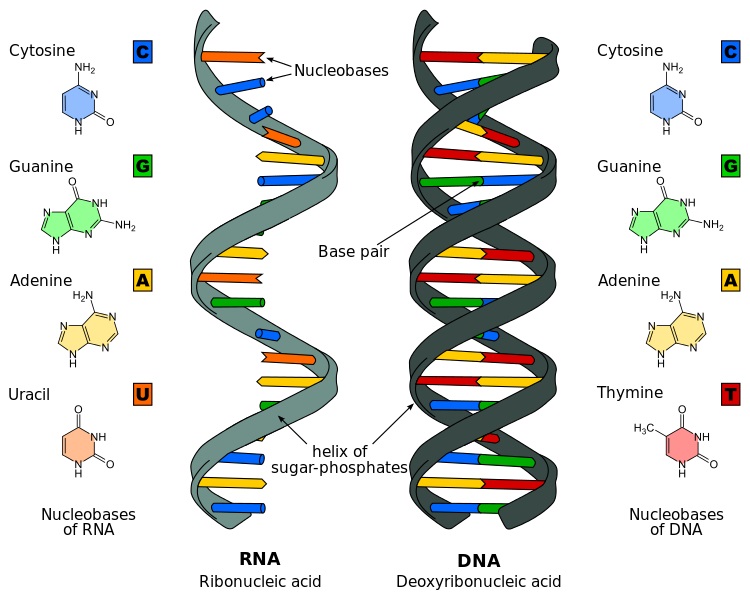
Nucleic Acids are Polymers of Nucleotides
DNA and RNA are both polymers made of individual nucleotides. The term “polymer” comes from “poly” for “many” and “mer” for parts, referring to the fact that each nucleic acid is made of many nucleotides.
Because nucleic acids can be made naturally by reacting inorganic ingredients together, and because they are arguably the most essential ingredient for life on Earth, some scientists believe that the very first “life” on Earth may have been a self-replicating sequence of amino acids that was created by natural chemical reactions.
Nucleic acids have been found in meteorites from space, proving that these complex molecules can be formed by natural causes even in environments where there is no life.
Some scientists have even suggested that such meteorites may have helped create the first self-replicating nucleic acid “life” on Earth. This seems possible, but there is no firm evidence to say whether it is true.
Nucleic Acid Structure
Because nucleic acids can form huge polymers which can take on many shapes, there are several ways to discuss the “structure of nucleic acid”. It can mean something as simple as the sequence of nucleotides in a piece of DNA, or something as complex as the way that DNA molecule folds and how it interacts with other molecules. Nucleic acids are formed mainly with the elements carbon, oxygen, hydrogen, nitrogen, and phosphorus.
Please refer to our Nucleic Acid Structure article for more information.
Monomer of Nucleic Acids
Nucleotides are the individual monomers of a nucleic acid. These molecules are fairly complex, consisting of a nitrogenous base plus a sugar-phosphate “backbone.” There are four basic types of nucleotide, adenine (A), guanine (G), cytosine (C), and thymine (T).
When our cells join nucleotides together to form the polymers called nucleic acids, it bonds them by replacing the oxygen molecule of the 3′ sugar of one nucleotide’s backbone with the oxygen molecule of another nucleotide’s 5′ sugar.
This is possible because the chemical properties of nucleotides allow 5′ carbons to bond to multiple phosphates. These phosphates are attractive bonding partners for the 3′ oxygen molecule of the other nucleotide’s 3′ oxygen, so that oxygen molecule pops right off to bond with the phosphates, and is replaced by the oxygen of the 5′ sugar. The two nucleotide monomers are then fully linked with a covalent bond through that oxygen molecule, turning them into a single molecule.
Nucleotides are the monomers of nucleic acids, but just as nucleic acids can serve purposes other than carrying information, nucleotides can too.
The vital energy-carrying molecules ATP and GTP are both made from nucleotides – the nucleotides “A” and “G,” as you might have guessed.
In addition to carrying energy, GTP also plays a vital role in G-protein cell signaling pathways. The term “G-protein” actually comes from the “G” in “GTP” – the same G that’s found in the genetic code.
G-proteins are a special type of protein that can cause signaling cascades with important and complex consequences within a cell. When GTP is phosphorylated, these G-proteins can be turned on or off.
Quiz
