Myofibril Definition
A myofibril is a component of the animal skeletal muscle. Myofibrils are long filaments that run parallel to each other to form muscle (myo) fibers. The myofibrils, and resulting myofibers, may be several centimeters in length. The muscle fibers are single multinucleated cells that combine to form the muscle. Myofibrils are made up of repeating subunits called sarcomeres. These sarcomeres are responsible for muscle contractions.
Myofibril Structure
Myofibrils are made up of two types of filaments: thin filaments and thick filaments. Thin filaments are composed of strands of the protein actin and a regulatory protein coiled together, whereas thick filaments are composed of strands of the protein myosin. The thin and thick filaments form partially overlapping layers that are laid out in functional units called sarcomeres. Because of the way the myofilaments are arranged, the myofibril appears to have dark and light bands, giving the muscles a striated appearance. The dark bands are known as A bands, and consist of thick filaments and some thin filaments. At the center of the A band is the H-zone, where only thick filaments are present, and the M-line, which contains enzymes involved in energy metabolism. The light bands, known as I bands, are the regions containing thin filaments only, and are found between the A bands. The I bands are centered on a region known as the Z line, a disk made up of the protein α-actinin that anchors the thin actin filaments and acts as a boundary between sarcomere subunits.
Myofibril Function
Myofibrils are made up of sarcomeres, the functional units of a muscle. The function of the myofibril is to perform muscle contraction via the sliding-filament model. When muscles are at rest, there is incomplete overlap between the thin and thick filaments, with some areas containing only one of the two types. When a muscle contracts, the sarcomeres shorten in length due to the thick and thin filaments sliding over each other, resulting in greater overlap between the filaments and a shortening of the H-zone and the I band. While the sarcomere length decreases during muscle contraction, the lengths of the myofilaments themselves do not change.
The myofilament movement is powered by the hydrolysis of ATP into ADP and inorganic phosphate. At rest, an ATP molecule is attached to a globular myosin head on the thick filament As the ATP is hydrolyzed, the myosin head changes conformation and forms an attachment known as a cross-bridge with the thin filament. When the ADP and phosphate molecules are released, the myosin head again changes conformation and pushes the thin filament towards the center of the sarcomere. When a new ATP molecule then binds to the myosin head, the head returns to its initial conformation and releases the thin filament in its new position nearer the central M-line. The cycle then repeats itself: the new ATP molecule is hydrolyzed into ADP and inorganic phosphate, and the myosin head changes conformation, resulting in the thin filament being pushed towards the center of the sarcomere. Each thick filament contains several hundred myosin heads that can form cross-bridges with the thin filaments about five times per second. The continuous contractions of the myofibrils result in a muscle contraction.
Muscle contractions are powered by ATP. As the muscle fiber itself only stores a very small amount of ATP, the energy comes from two other compounds stored in the muscles: creatine phosphate and glycogen. The ATP stored in a muscle fiber, and the ATP that can be formed by creatine phosphate, are used for short-term bursts of energy; they can provide energy for up to about 15 seconds. Glycogen can provide a long-term energy source as glycogen breaks down to glucose, which is then converted to ATP through glycolysis and aerobic respiration.
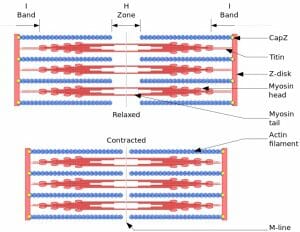
This figure depicts a sarcomere, a unit within a myofibril. It shows the thin actin filaments and the thick myosin filaments, and how their positions change as a muscle contracts.
Quiz
1. How many types of myofilaments are found in a myofibril?
A. 1
B. 2
C. 4
D. none
2. What structure acts as a boundary between sarcomeres?
A. A-band
B. H-zone
C. M-line
D. Z-line
3. What is the best long-term energy source for muscle contraction?
A. ATP
B. starch
C. glycogen
D. creatine phosphate
References
- Campbell, N. A., & Reece, J. B. (2005).Biology, 7th. ed. Ch. 49. San Francisco, CA: Benjamin Cummings. ISBN: 0-8053-7171-0.
- Randall, D., Burggren, W., & French, K. (2002).Eckert animal physiology: mechanisms and adaptations, 5th. ed. Ch. 10. New York, NY: W.H. Freeman and Company. ISBN: 0-7167-3863-5.
