Cytotoxic T cells (AKA ‘killer’ T cells) are key lymphocytes of the adaptive immune system. Like helper T cells, they are activated by antigen-presenting cells in the lymph nodes. However, whereas helper T cells mediate the activity of other immune cells against a pathogen, cytotoxic T cells directly attack and destroy infected host cells.
What is a Cytotoxic T Cell?
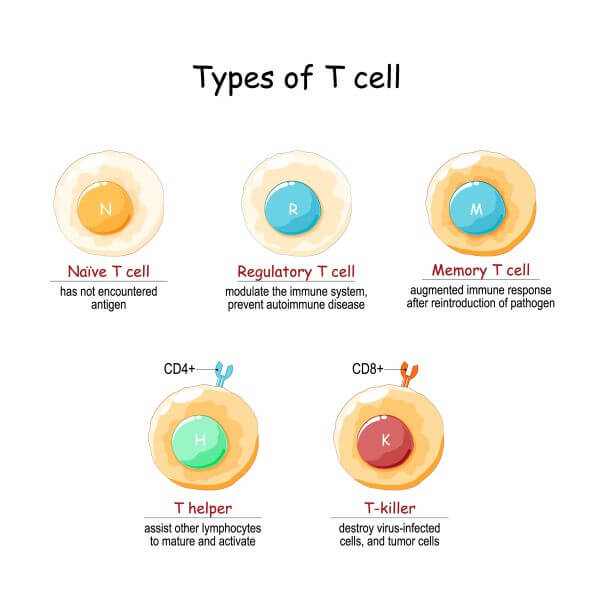
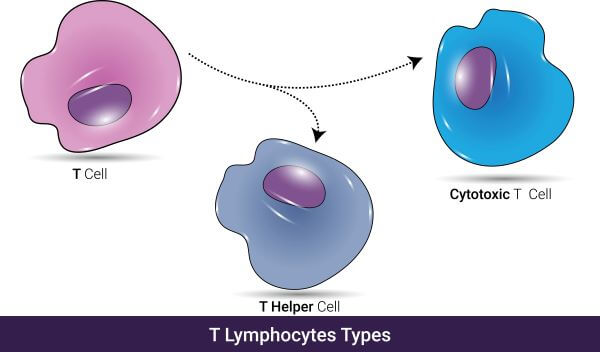
Cytotoxic T cells and helper T cells are the two major types of T lymphocytes in the human immune system. Both originate from stem cells in the bone marrow before migrating to the thymus, where they mature and differentiate into CD4+ and CD8+ T cells.
All T cells express T cell receptors (TCRs) on their surface, and they also express CD4 or CD8 co-receptors, depending on their function. Helper T cells express CD4 receptors (and may be referred to as CD4+ T cells), and cytotoxic T cells express CD8 receptors and are also known as CD8+ T cells.
Helper T cells ‘help’ to coordinate the adaptive immune response by activating other cells of the immune system, whereas cytotoxic T cells directly kill infected cells.
Activation of Cytotoxic T Cells
All T cells are considered ‘naïve’ until they encounter their specific antigen. Like helper T cells, cytotoxic T cells are specific to just one type of antigen. However, they cannot bind to their antigen directly; instead, they rely on assistance from antigen-presenting cells (APCs).
APCs (such as dendritic cells, macrophages, and B cells) find and engulf invading pathogens before digesting them into protein fragments. Some of these fragments are displayed on their surface as part of a structure called the Major Histocompatibility Complex (MCH) and presented to T cells in the lymph nodes. When a cytotoxic T cell encounters its specific antigen on the surface of an APC, its TCR binds to the MHC and the T cell is activated.
There are two types of Major Histocompatibility Complexes found on the surface of APCs; these are MCHI and MCHII. The key difference in the activation of helper T cells vs. cytotoxic T cells is that they each bind to a different type of MCH receptor. Cytotoxic T cells bind to MHCI complexes, whereas helper T cells can only bind to MHCII complexes.
Once a naïve T cell has been presented with its antigen, it rapidly divides and cytotoxic T cells begin to migrate to areas of infection in the body.
Function of Cytotoxic T Cells
Cytotoxic T cells are also known as ‘killer’ T cells thanks to their role in the destruction of infected cells, pathogens, and tumor cells. The main way they do this is vis the transfer of cytotoxic granules to infected target cells, which kill the cell and any pathogens it contains.
Secretion of Cytotoxic Granules
Cytotoxic T cells and natural killer (NK cells) use a very similar mechanism for the destruction of virus-infected cells and pathogens.
Like NK cells, cytotoxic T cells contain cytotoxic granules containing proteins called perforin and granzymes, which work together to destroy target cells. When they encounter an infected cell, cytotoxic T cells bind to the MHCI via their TCR receptors and release their cytotoxic granules. Perforin creates holes in the cell membrane of the target cell, and granzymes enter the cell via these pores. Once inside, the granzymes initiate apoptosis (programmed cell death) which kills the cell and any pathogens it contains.
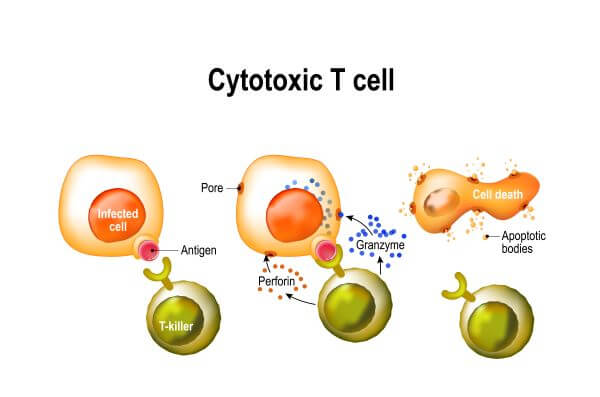
Cytotoxic T cells typically target virus-infected cells, so this method of killing benefits the host in that it contains and destroys the viruses within the cell. If the cell were simply lysed, the viruses could spill out of the cell and migrate to other parts of the body.
The main difference between the action of NK cells and cytotoxic T cells is in how they recognize their targets. Natural killer cells are stimulated to attack by the absence of MHCI, which is typically lost from the surface of tumor cells and pathogen-infected cells. Cytotoxic T cells, however, recognize their targets by the presence of specific antigens in the MCHI complex. Both cytotoxic T cells and NK cells are ‘serial killers’ and can bind to and attack multiple target cells.
Secretion of Cytokines
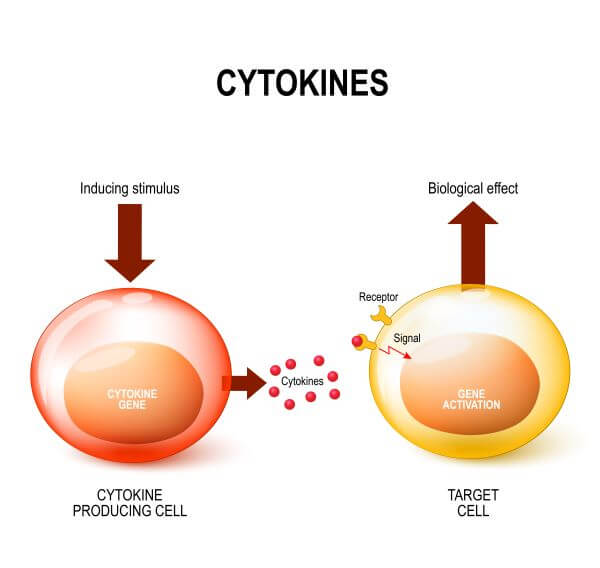
Perforin and granzyme secretion is the main weapon of the cytotoxic T cell, but it isn’t their only function. Cytotoxic T cells also secrete cytokines, which contribute to the adaptive immune response in a number of ways.
One of these cytokines (IFN-γ) directly inhibits viral replication, which helps to slow the spread of infection in the body. IFN-γ also activates macrophages and stimulates them to migrate to sites of infection, where they function both as effector cells (as they engulf and destroy pathogens) and as antigen-presenting cells.
Regulation of Cytotoxic T Cells
Cytotoxic T cells are key players in the adaptive immune response and are highly effective in clearing infected cells from the body. However, if left unchecked, cytotoxic T cells can contribute to an excessive immune response, resulting in damage to healthy host cells and tissues.
In most cases, this is prevented by an event known as the contraction phase, which takes place a few weeks after the initial infection. Cytotoxic T cell activity typically peaks around 7 days after the initial infection. In the days and weeks following the peak of cytotoxic T cell activity, the contraction phase begins and up to 95% of the cells die off. The remaining 5-10% become memory T cells, which remain in the body for a long time after the infection has been cleared.
What is a Memory Cytotoxic T Cell?
Memory cytotoxic T cells are the small percentage of cytotoxic T cells that remain in the body following the contraction phase. These cells persist in the immune system for a long time, and quickly recognize and launch an attack against the pathogen in the event of re-infection.