Definition
The red blood cell or erythrocyte is an unusual, unique, and highly differentiated cell without organelles or the ability to divide. The erythrocyte is central to body physiology as it is responsible for oxygen transport through the bloodstream. Produced in the bone marrow from hematopoietic stem cells, red blood cells live for approximately 120 days. They can communicate and interact with endothelial cells, blood platelets, macrophages, and bacteria via membrane proteins.
Red Blood Cell Structure
Red blood cell structure is very simple compared to other cell types as they have no organelles. The most common blood cell type, these biconcave cells have no organelles.
A mature red blood cell is an anucleate cell – it has no nucleus. This means it contains no DNA. Red blood cell size is around eight thousand nanometers (eight micrometers) in diameter. It has a unique biconcave shape; the cell can move easily through the smallest capillaries but the biconcave shape produces a large surface area.
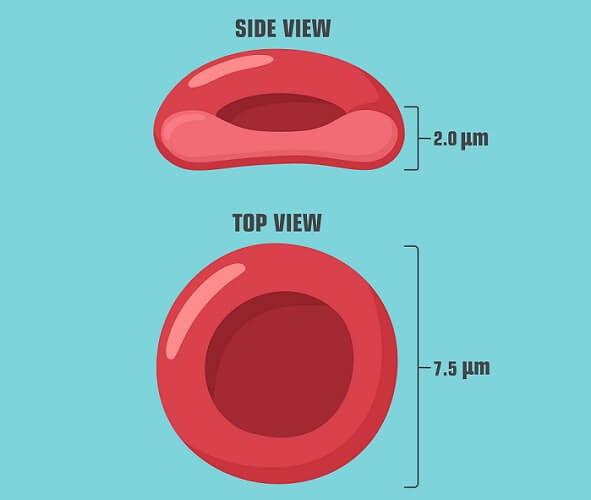
A lipid bilayer membrane covers the cell’s cytoplasm. This cytoplasm contains no organelles but high levels of hemoglobin – a protein formed from four polypeptide chains (globins) and four carbon-based heme molecules that each surround one iron ion. There are two main types of hemoglobin – HbA (adult hemoglobin) and HbF (fetal hemoglobin).
The RBC membrane contains integral and peripheral proteins. Integral proteins distinguish individuals in the form of A, B, O, and AB blood types. They also support the internal structure and bind hemoglobin. Peripheral membrane proteins are found on the inner side of the membrane and help to make the red blood cell extremely elastic.

Red Blood Cell Development
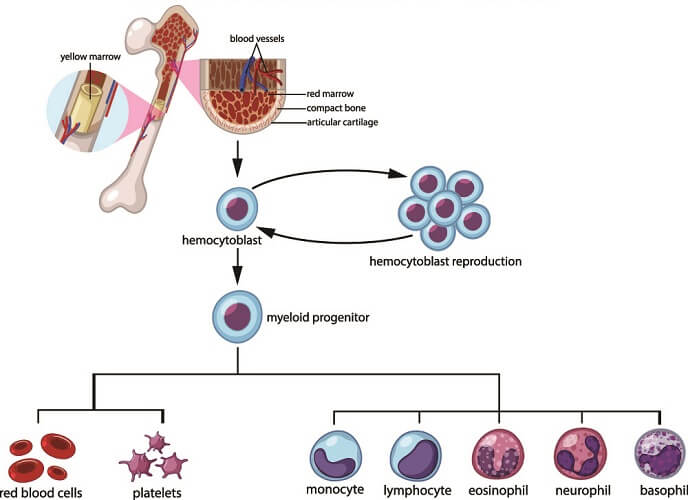
The development of erythrocytes – called erythropoiesis – begins in the red bone marrow. The process produces around two million red blood cells (RBCs) every second and the time required from multipotential myeloid stem cell to red blood cell takes about two days. While red blood cell production most commonly occurs in the red bone marrow, some disorders lead to extramedullary erythropoiesis in the liver, thymus, and/or spleen – the areas where fetal red blood cells are produced.
Multipotential hematopoietic stem cells (hemocytoblasts) in the red bone marrow differentiate into a common myeloid progenitor cell that can either become a thrombocyte (blood platelet) or an erythrocyte. The myeloid progenitor is encouraged to produce proerythroblasts – the first form of a true red blood cell – by chemical messengers. While a myeloid progenitor cell is an oligopotent stem cell that can turn into two related types of cells, the proerythroblast can only ever become an RBC. It is, therefore, a unipotent stem cell. This immature cell type has a large nucleus, cytoplasm, and ribosomes. It can divide via mitosis.
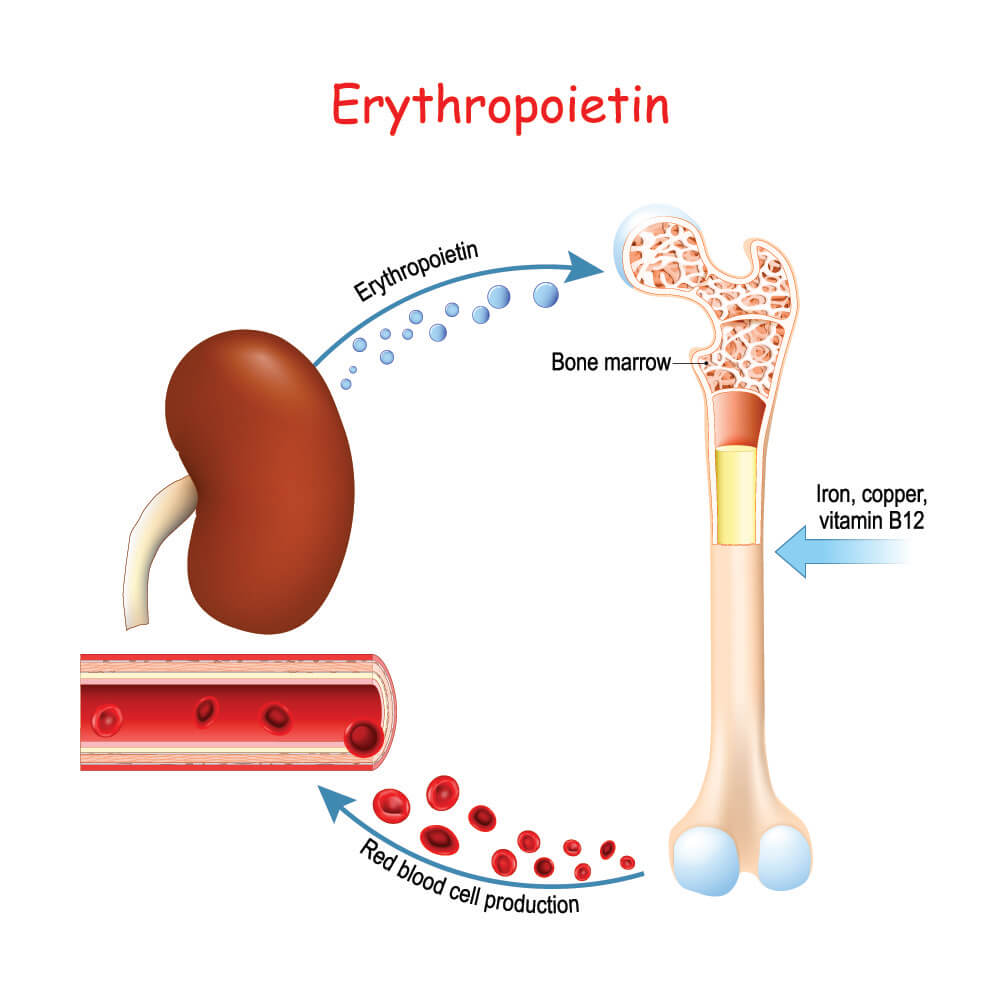
When the kidneys detect low oxygen levels, they secrete the cytokine hormone erythropoietin. This causes common myeloid progenitor cells in the bone marrow to differentiate into proerythroblasts.
Erythropoiesis is strictly regulated by erythropoietin and stem cell factor (SCF). They produce a normal red blood cell count of 4.2 to 6.1 million cells per microliter of blood. When the kidneys detect low oxygen levels, stress erythropoiesis takes place. In combination with SCF, RBC survival, differentiation, and proliferation (growth) rates are increased. Recent studies have shown that a fragment of ribonucleic acid – miR-451 – also plays a major role in red blood cell maturation in mice. It is thought that micro-RNA allows the maturation process to continue in humans, too.
Red blood cells formed in the bone marrow are immature. The formerly-described proerythrocytes differentiate into basophilic erythroblasts here. This page shows images of the different stages of red blood cell development and, as you will see, the nucleus is very much visible in the basophilic erythroblast, although the cell has shrunk. The nucleolus is lost during this change and the cytoplasm is still packed with ribosomes.
The basophilic erythroblast then differentiates into a polychromatophilic erythroblast – an even smaller cell. A color change occurs as the ribosomes in the cytoplasm start to produce more hemoglobin. The nucleus shrinks a little more. The nucleus to cytoplasm ratio is 60 – 80%.
Remaining in the red bone marrow, the next step of red blood cell maturation is the differentiation from polychromatophilic to orthochromatophilic erythroblast. The cell and its nucleus shrink again; the nucleus begins to degrade.
The final stage of maturation in the bone marrow is the step from orthochromatophilic erythroblast to reticulocyte. A reticulocyte is around half the size of the proerythroblast and has no nucleus. Hardly any more ribosomes exist as the cell now contains enough hemoglobin protein to function (see next heading). The smaller size allows the reticulocyte to exit the bone marrow and enter the bloodstream.
A red blood cell matures in the blood. A mature RBC is about two microns smaller in diameter than the preceding reticulocyte. It has no organelles and is merely composed of a lipid membrane with surface proteins, cytoplasm, and protein (hemoglobin).
Red Blood Cell Function
RBC function is to transport oxygen and (in much smaller quantities) carbon dioxide between the tissues of the body and the lungs. Oxygen and carbon dioxide can bind to the iron ions that sit inside the hemoglobin.
As each hemoglobin molecule contains four iron ions, in theory, each red blood cell transports four oxygen or four carbon dioxide molecules. Oxyhemoglobin is hemoglobin bound to oxygen. Carbaminohemoglobin is hemoglobin bound to carbon dioxide.
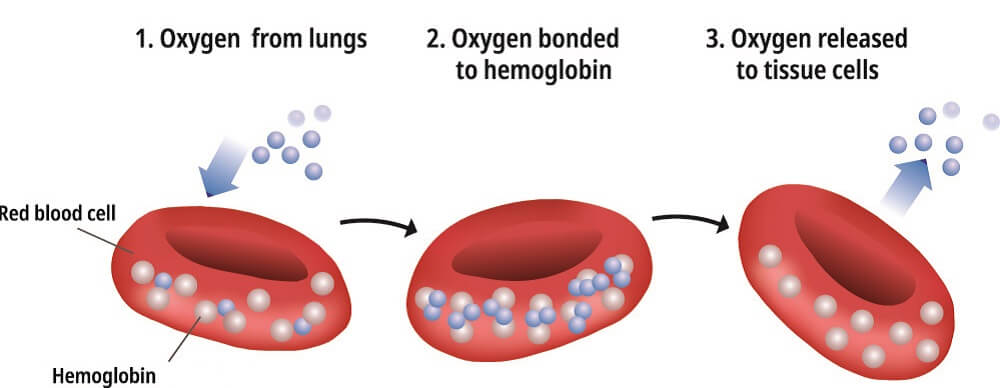
Oxygen Transport
The initial attachment of one oxygen molecule (O2) to hemoglobin is relatively difficult as the shape of the hemoglobin makes a first connection complicated. Only after the first oxygen molecule has bound to the first iron ion and the shape of the hemoglobin changes does the binding of additional oxygen become easier. However, after the addition of a third oxygen molecule, the shape is no longer so efficient; a fourth oxygen will not bind as easily. The more oxygen molecules attached to a hemoglobin protein, the brighter red it becomes.
How much oxygen is transported by the red blood cell depends on many variables. Naturally, the presence of oxygen in the blood and lung function are most important. Blood acidity (pH), body temperature, unbound carbon dioxide levels (that make the blood more acidic), and certain genetic disorders can all affect the ability of RBCs to carry enough oxygen to the tissues. Blood vessel damage does not affect hemoglobin’s affinity for oxygen but can stop erythrocytes from reaching certain areas.
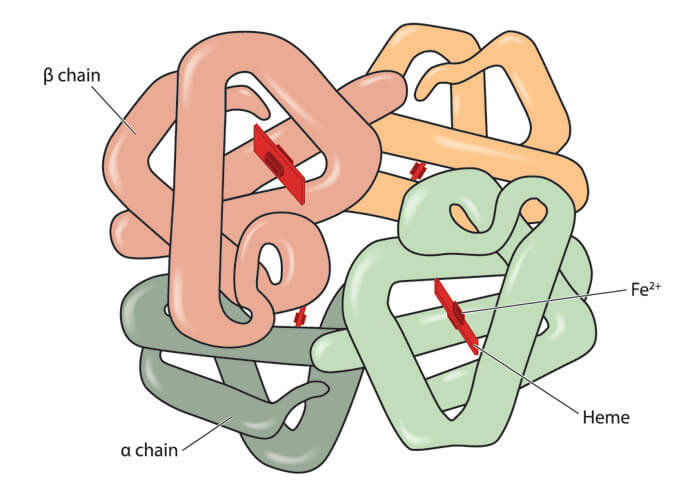
One erythrocyte can contain anywhere up to 300 million hemoglobin molecules. This means that every mature, healthy cell can transport more than one billion oxygen molecules.
Carbon Dioxide Transport
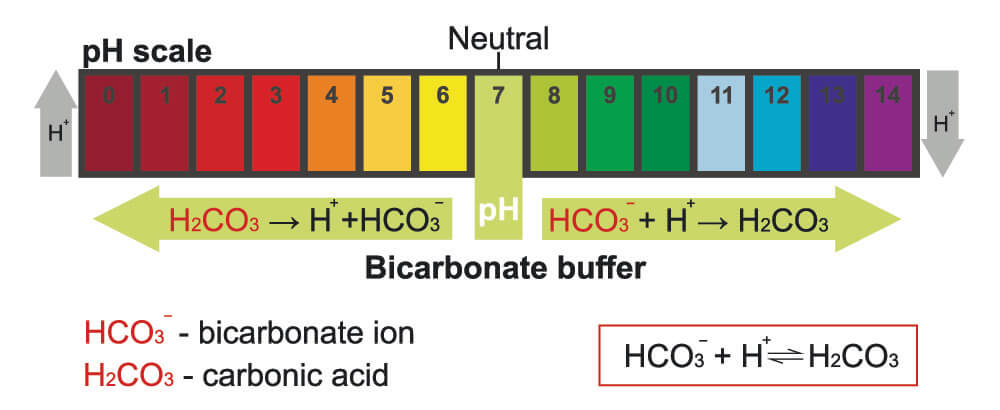
While 98% of our oxygen is transported via the blood, less than 10% of carbon dioxide (CO2) binds to hemoglobin. The rest is converted (within the red blood cell) into carbonic acid. This occurs because red blood cell cytoplasm contains a converting enzyme called carbonic anhydrase. Carbonic acid is very unstable and forms positively-charged hydrogen ions and negatively-charged bicarbonate ions. These ions form the basis of the bicarbonate buffer system that controls body acidity (pH).
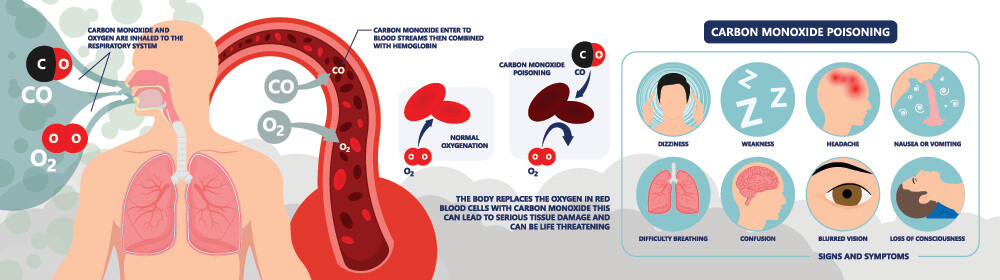
Carbon Monoxide Transport
Carbon monoxide poisoning usually occurs in closed-off areas with open gas heating systems. Carbon monoxide (CO) is not usually present in the body in significant quantities. However, if available it is more likely to bind to hemoglobin than oxygen. Carbon monoxide, therefore, blocks oxygen transport; a person will slowly suffocate should they remain in a toxic area. Treatment is the rapid administration of 100% oxygen.
The Red Blood Cell Test
The red blood cell count is part of the CBC (complete blood count) that gives any physician an immediate indication of a person’s general level of health.
Gender and age determine what is considered a high RBC count, normal RBC count, or low RBC count:
- Men: normal – 4.7 to 6.1 million red blood cells per microliter (cells/mcL)
- Women: normal – 4.2 to 5.4 million cells/mcL
- Children: normal – 4.0 to 5.5 million cells/mcL
Low red blood cell count causes include blood loss, anemia, certain vitamin deficiencies that lower RBC proliferation rates, kidney disease (low levels of erythropoietin), and genetic disorders that affect either protein synthesis or red blood cell maturation and lifespan regulation.
High red blood cell count causes include chronic low oxygen levels (smoking, living in areas of high elevation, heart disease, lung conditions), dehydration (this affects the ratio of blood cells to blood plasma), and genetic or inherited disorders such as familial erythrocytosis and polycythemia.

Other RBC tests include:
- Hematocrit: measures the volume of RBCs in the total blood (cells and plasma) as a volume percentage.
- HbA1C: not only O2, CO2, and CO bind to hemoglobin. Glucose can do this, too. The glycated hemoglobin test looks at how much glucose is attached to hemoglobin. Glucose does not attach to the iron ions and so does not affect oxygen transport.
- ABO typing: tests the compatibility of red blood cell integral proteins to ensure typed blood transfusions are safe.
- Erythrocyte sedimentation rate (ESR): measures how quickly RBCs move through the plasma. This can help to diagnose inflammatory illnesses.
- Red blood cells in urine: hematuria, or blood in the urine, can be gross (visible) or microscopic (only visible under a microscope). This can indicate urinary tract infection, kidney stones, polycystic kidney disease, urinary tract cancers, or traumas. Some blood-thinning medications also cause RBCs in urine.