Ionizing Radiation Definition
The term radiation means to give off energy as waves or particles. Ionizing radiation gives off energy by knocking electrons off atoms, which causes the atoms to have a charge. Another term for a charged particle is an ion. The charges on the atomic particles make ionizing radiation unstable and reactive. The particles radiate because they are trying to stabilize themselves. Ionizing radiation is a high-energy type of radiation because it comes from the high-energy end of the electromagnetic spectrum. The creation of ionizing radiation is a natural process in the universe.
Sources of Ionizing Radiation
Ionizing radiation comes from these main sources:
- Nuclear reactions in the Earth’s sun and stars in space
- Radioactive decay in the body’s tissues and in the soil
- Radioactive decay of unstable elements in rocks, especially rocks that contain radium and release radon gas
- Occupational sources from mining, medical, research and industrial jobs.
Types of Ionizing Radiation
Types of ionizing radiation include:
- x-rays/gamma rays
- alpha particles
- beta particles
- neutron particles
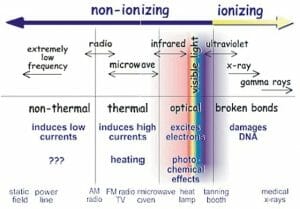
The image above shows where ionizing radiation is located in the electromagnetic spectrum.
Examples of Ionizing Radiation
X-Rays/Gamma Rays
X-rays and gamma rays travel in the form of energy “packets” called photons. Just like light, photons travel in a straight line. X-rays come from a change in energy levels in an atom. Medical x-rays show bones and other dense structures in the body because these structures absorb the radiation; soft tissues like skin and organs don’t. Gamma rays are photons that come from the nucleus of the atom. Both types of ionizing radiation can pass through the human body if they have enough energy. This radiation can destroy whole cells and/or damage the DNA in the cell’s nucleus, which can lead to DNA mutations and the possibility of cancer.
Alpha Particles
Alpha particles don’t penetrate skin as x-rays and gamma rays do. In fact, a piece of paper or fabric can stop alpha particles. These particles are heavier and slower than x-rays and gamma rays because they have two protons and two neutrons in their nuclei. However, these particles become dangerous when they are inhaled. Radon is a colorless, tasteless and odorless gas which comes from the decay of the element radium. Radium occurs naturally in the Earth’s rocks and soil, and is made primarily of alpha particles, so inhaling radon puts alpha particles directly in contact with the lung tissue. The alpha particles from radon (when inhaled) are about 20 times as effective as x-rays and gamma rays at causing cancer.
Beta Particles
Beta particles are electrons which are much smaller and faster than alpha particles. Most solid objects stop them, but they can easily penetrate human skin and cause tissue damage and burns. The severe burns suffered by people exposed to the fallout of nuclear reactor explosions and atomic bombs are due to beta particles. These burns are also called beta burns. Beta particles can be ingested or inhaled if they contaminate water and food supplies, and ingestion causes even more severe damage. However, very small amounts of some kinds of beta particle-emitting elements (like iodine 131) are used in medicine to diagnose and treat diseases of the thyroid gland.
Quiz
1. Beta particles __________________.
A. are big and slow
B. come from the sun
C. are small and fast
D. have two protons and two neutrons in their nuclei.
2. Radon gas has mostly which type of particles?
A. beta
B. x-ray
C. alpha
D. neutron
3. Which of the following is NOT a source of ionizing radiation?
A. fire
B. rocks
C. the sun
D. x-rays
References
- Doss, H. (2017, May 14). Ionizing radiation and humans – the basics. Retrieved from http://physicscentral.com/explore/action/radiationandhumans.cfm.
- Grabianowski, E. (2017, May 14). How radiation sickness works. Retrieved from http://science.howstuffworks.com/radiation-sickness1.htm.
- Ionizing radiation. (n.d.). In Encyclopedia Britannica online. Retrieved from https://www.britannica.com/science/ionizing-radiation.
- What is ionizing radiation? (2017, May 14). Retrieved from https://biotech.law.lsu.edu/research/reports/ACHRE/intro_9_1.html.
