Condensation Definition
Condensation has multiple meanings in the field of biology. A condensation reaction is when two smaller molecules join to form a larger one by removing functional groups that form a small molecule, often water. Condensation can also refer to a state change in water from gas to liquid, which is an important step in the water cycle. There is also DNA condensation, which is when chains of deoxyribonucleic acid (DNA) compact into a denser structure when a cell is preparing to divide.
Examples of Condensation
Condensation Reactions
A condensation reaction is the formation of a larger molecule from two smaller ones, which also form another, smaller molecule by losing functional groups in order to join together. Some examples of small molecules that are byproducts of condensation reactions are acetic acid (CH3COOH) and methanol (CH3OH), but most often water (H2O) is formed from one molecule losing hydrogen (H) and another losing a hydroxyl group (-OH), especially in the biological reactions that take place in living things. When water is a product, the condensation reaction is often called a dehydration reaction.
Condensation reactions are used to make crucial large molecules called macromolecules in the body, such as carbohydrates, lipids, and proteins. Carbohydrates are simple sugars such as glucose, which is used for energy in the body. A sugar with one ring is a monosaccharide, and condensation reactions join units of glucose together to form disaccharides, with water also being produced as a result. When more than two sugars are joined, the molecule is called a polysaccharide. Glucose is stored in long chains that are made through many dehydration reactions, and this molecule is called glycogen. When more energy is needed in the body, glucose is one necessary component, and glucose molecules are then broken off of the chain by hydrolysis, the addition of water (and the opposite of dehydration reactions).
Similarly, glycerol and three fatty acids are joined together through condensation reactions to make a lipid molecule, and amino acids are joined to form proteins. In the body, lipids are fats and certain vitamins, and they have many functions such as energy storage, cell signaling, and forming the lipid bilayer of the cell membrane. Proteins help certain chemical reactions to occur and also have roles in cell signaling and providing structure to parts of the body; for example, the proteins collagen and elastin are found in cartilage. Without condensation reactions, all of these important molecules could not be formed.
Condensation in the Water Cycle
Condensation is part of the water cycle, which describes how water continuously cycles throughout the Earth in solid, liquid, and gaseous forms. During condensation, water vapor in the air changes from a gas to liquid water. As altitude increases, temperature and water pressure both decrease, which causes water vapor to turn into liquid water. A very large concentration of these droplets of water forms clouds and fog. The water then falls to Earth’s surface as precipitation. It falls back into the Earth’s waterways and also enters the soil, where it can be absorbed by plants. Water can also freeze into its solid form, ice, and then melt back into water. This can occur as snow or on the Earth’s surface where temperatures are below freezing. Next, evapotranspiration occurs, and the water changes from the liquid from to the gas form. Evapotranspiration is the combination of evaporation from Earth’s oceans, lakes and rivers and transpiration, which is when excess water evaporates from plants. Evapotranspiration allows water to enter back into the air, and then the water cycle starts all over again. The water cycle is crucial in all of Earth’s ecosystems because all living things need water to survive.
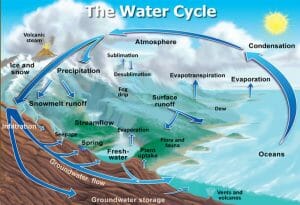
This is a diagram of the components of the water cycle.
DNA Condensation
DNA condensation occurs in all organisms. Unlike the other forms of condensation mentioned in this article, this condensation does not involve water; it involves making DNA fit into a smaller space. In eukaryotes, which include all living things that are not archaea or bacteria, DNA condensation occurs during mitosis, the process of cell division. During the prophase stage of mitosis, DNA strands condense into nucleosomes, which are segments of DNA wrapped around histone proteins. DNA wrapped around proteins is called chromatin. Nucleosomes are then folded even more to form chromosomes. DNA is always folded to some extent; if all the DNA in one of your cells were stretched out, it would be 2 meters long!
Bacterial DNA forms a circular, continuous loop. In order for all of a bacterium’s DNA to fit within it, the DNA strands must be made about 1000 times smaller. Instead of histone proteins in eukaryotic cells, DNA binding proteins are used to form loops within the circular chromosome to condense it. Then, these loops twist into themselves to condense the DNA even further, in a process known as supercoiling.
Even viruses, which are not considered true living things because they cannot reproduce without invading a cell, have DNA that undergoes condensation. The DNA must be condensed because it is longer than the virus’s capsid, which is the protein shell that contains the DNA. The DNA is essentially wound like a spool inside the capsid by a powerful motor that can condense DNA very tightly.
Related Biology Terms
- Dehydration – The removal of water; dehydration reactions remove water from two molecules as they are joined to form a larger one.
- Water cycle – The flow of water in all its forms throughout the Earth, its organisms, and the air.
- DNA condensation – The winding of DNA so that it can fit into a small space.
- Mitosis – The process of cell division in eukaryotic cells.
Quiz
1. Which is a type of condensation?
A. Water vapor becoming liquid water
B. The joining together of glucose molecules into a disaccharide
C. The packing of DNA into a small space
D. All of the above
2. What phase change does water undergo during condensation in the water cycle?
A. Liquid to gas
B. Gas to liquid
C. Liquid to solid
D. Solid to gas
3. What is DNA wrapped around during condensation in eukaryotic cells?
A. Histone proteins
B. DNA binding proteins
C. Chromatin
D. Nucleosomes
