Cofactor Definition
A cofactor is a non-protein chemical that assists with a biological chemical reaction. Co-factors may be metal ions, organic compounds, or other chemicals that have helpful properties not usually found in amino acids. Some cofactors can be made inside the body, such as ATP, while others must be consumed in food.
Minerals, for example, come from the environment, and cannot be made from scratch by any living cell. The organic compounds we refer to as “vitamins” are cofactors that our own bodies cannot make, so we must consume them from food in order for our cells to be able to perform essential life functions.
At the biochemical level, cofactors are important in understanding how biological reactions proceed. The presence or absence of cofactors may determine how quickly reactions proceed from their reactant to their product.
At the biological level, understanding cofactors is important to understanding health. Without the proper cofactors, humans and other animals can develop serious diseases and even death.
Function of Cofactors
Cofactors generally serve the purpose of supplying chemical groups or properties that are not found in other chemical groups.
ATP, for example, is a cofactor with a unique ability to transfer energy to drive chemical processes such as the activity of enzymes and transport proteins.
Heme, on the other hand, is a chemical complex that contains iron, which allows heme to bond to oxygen molecules in a unique way. Heme is necessary for our blood cells to carry oxygen through our bodies.
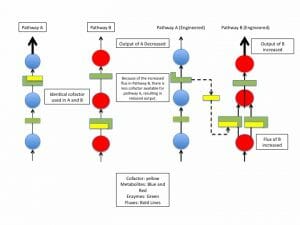
There are dozens of known cofactors, each of which may be necessary for multiple biochemical reactions, as illustrated below.
As a result, the functions of cofactors may be as diverse as their chemical structures and properties.
The wide-ranging effects of cofactors can be seen by studying vitamin deficiencies: deficiencies of different vitamins, many of which are cofactors, have many different negative effects on human health.
Types of Cofactor
Vitamins
Vitamins are organic compounds that are co-factors for necessary biochemical reactions. Vitamins typically need to be consumed in the diet, because they cannot be made inside the body.
Many vitamins are cofactors which help enzymes to catalyze reactions, such as the production of important proteins. Vitamin C, for example, is a cofactor for the production of the connective tissue collagen.
This is why people who get scurvy – a severe form of vitamin C deficiency – may experience connective tissue problems, including muscle weakness, muscle soreness, and even unexplained bleeding as the connective tissues of blood vessels cannot be replaced.
Vitamin deficiencies are a good illustration of the effects of co-factor deficiency. Just as there are many possible vitamin deficiencies with many different symptoms, there are many different co-factors that our body needs to carry out its diverse necessary biochemical reactions.
The body’s requirement for diverse vitamin cofactors is also the reason why nutritionists counsel people to “eat the rainbow” – many plants’ colors are produced by cofactors, so by eating fruits and vegetables in a wide variety of colors helps to ensure that we consume a healthy variety of cofactors.
Minerals
Like vitamins, minerals are chemicals from outside of the body that must be ingested to allow our cells to function properly. The difference is that while vitamins are organic molecules – molecules containing carbon, which are often made by other living things – minerals are inorganic substances that occur naturally, and are often found in rocks and soil.
Minerals often enter our diets from plants, which draw them up out of the ground through their roots along with water. In some rare cases, people with vitamin deficiencies may feel the urge to eat certain types of soil to obtain the minerals from the soil directly.
Minerals that are important for human health include copper, which is necessary for the function of some important liver enzymes that break down toxins; iron, which is necessary for the function of some important metabolic enzymes; magnesium, which is necessary for the function of DNA polymerase and other enzymes; and zinc, which is also necessary for DNA polymerase as well as some liver enzymes.
As with vitamins, there can be too much of a good thing – while minerals are necessary in small amounts for our metabolisms to function, taking large doses of them can result in toxicity and death. Indeed, overdoses of iron-containing multivitamins are a leading cause of death in children under 4, who may mistake these multivitamins for candy.
Organic Non-Vitamin Cofactors
Some cofactors are organic substances not classified as enzymes. Some of these may be made inside our own bodies, and so not qualified as vitamins.
Organic non-vitamin cofactors include ATP – an essential assistant to many biochemical processes, which transfers energy to numerous enzymes, transport proteins, and more; coenzyme Q, which plays a vital role in the mitochondrial transport chain; and heme, which is a complex iron-containing compound that is necessary for our blood cells to carry oxygen throughout our bodies.
Examples of Cofactors
Thiamine (Vitamin B3)
Thiamine is a vitamin found primarily in edible seeds such as beans, corn, and rice. To improve public health, thiamine is often artificially added to wheat-containing products such as breakfast cereals.
In the body, thiamine is used to make many co-enzymes that assist with important processes. It is made into thiamine pyrophosphate, which is necessary to break down sugars and amino acids.
Severe thiamine deficiency is one cause of Korsakoff Syndrome – a rare neurological disorder seen in people with severe alcohol addiction. In Korsakoff Syndrome, severe malnutrition, lack of thiamine, and brain damage from overuse of alcohol combine to produce severe symptoms, including memory impairment. Some sufferers of Koraskoff Syndrome are not able to form new memories because the metabolism of their brain is so impaired.
Folic Acid (Vitamin B9)
Folic acid is another vitamin which is now often added to food to improve public health. It is necessary for the body to produce DNA, RNA, and amino acids, which are necessary for growth and cell division.
This makes folic acid particularly essential for pregnant women, whose fetuses are producing new cells and tissues very quickly. Deficiencies in folic acid can lead to birth defects in babies, or to anemia in pregnant women who may not be able to make enough new blood cells to supply both them and the baby.
For this reason, it is recommended that all women of childbearing age talk to their doctors about taking folic acid supplements. Pregnancy outcomes are best when sufficient folic acid is present in the mother’s body even before pregnancy begins.
Iron-Sulfur Clusters
Iron-sulfur clusters are – you guessed it – clusters of iron and sulfur ions which can form stable arrangements. These clusters have many unique properties that are not found in amino acids or other organic compounds.
The unique properties of iron-sulfur clusters make them very useful for biological reactions involving electron transfers. Both iron and sulfur are able to store and release electrons with greater eases than more common atoms such as carbon.
This makes iron-sulfur clusters a vital part of cofactors and enzymes involved in electron transfer and energy transfer, including NADH dehydrogenase, coenzyme Q, cytochrome C, and Complex I and Complex II in the mitochondria.
Related Biology Terms
- Enzyme – A protein which increases the rate of a chemical reaction. Enzymes make life possible by catalyzing reactions that would otherwise proceed very slowly.
- Mineral – A naturally occurring inorganic substance, often found in rocks. Some minerals have chemical properties that are used by cells to facilitate their life processes.
- Vitamin – An organic compound that is essential for biological activity. Vitamins must be consumed in an organism’s diet if the organism cannot manufacture them themselves.
Quiz
1. Which if the following is NOT likely to be a cofactor?
A. Vitamin A
B. Iron
C. ATP
D. None of the above.
2. Why might a mineral be useful as a cofactor?
A. Minerals can have very different chemical properties from organic compounds such as carbon.
B. Some minerals are better at accepting and donating electrons than organic compounds.
C. Minerals include atoms that cannot be produced by living things, but most be obtained from the environment.
D. All of the above.
3. Why is it important to eat a variety of fruits and vegetables?
A. Because fruits and vegetables contain minerals which they take up from the soil through their roots.
B. Because fruits and vegetables contain organic compounds which animals do not make themselves.
C. Because different fruits and vegetables include different cofactors that are necessary to human health.
D. All of the above.