Definition
Glutamic acid (Glu or E) is an amino acid with the molecular formula C5H9NO4. It is a non-essential amino acid, meaning it can be synthesized by the body.
Under physiological conditions, the carboxyl group of glutamic acid has lost a proton, producing a negative charge. This form of the amino acid is called glutamate, and this is the form that is abundant in the human body.
Glutamic acid is an essential building block of proteins. Independent of its role as an amino acid, glutamate is also a vital signaling component in the central nervous system and is highly abundant in the brain.
Structure and Properties of Glutamic Acid
Glutamic acid is an amino acid with the molecular formula C5H9NO4. Its symbols are either Glu or E. As with all amino acids, it has a carboxyl-terminal end, an amino-terminal end, and a side chain. The side chain of glutamic acid has a carboxylic acid group. In the genetic code, the mRNA codons that encode for its incorporation into a polypeptide chain are GAA and GAG.
As with all the amino acids (except glycine), glutamic acid has two forms: an L-form and a D-form. These forms are stereoisomers, differing only in the spatial arrangement of their atoms. Usually, only the L-form is found in cells. Consistently, glutamic acid is found almost exclusively in its L-form, except in some special cases. For example, the D-form is found in the cell wall of some bacteria and in the cells of the liver.
Glutamate cannot cross the blood-brain barrier, so even if it is obtained through the diet, it also has to be synthesized in the brain. It is synthesized from α-ketoglutarate, which is an intermediate molecule generated during the Krebs Cycle.
Glutamic Acid vs. Glutamate
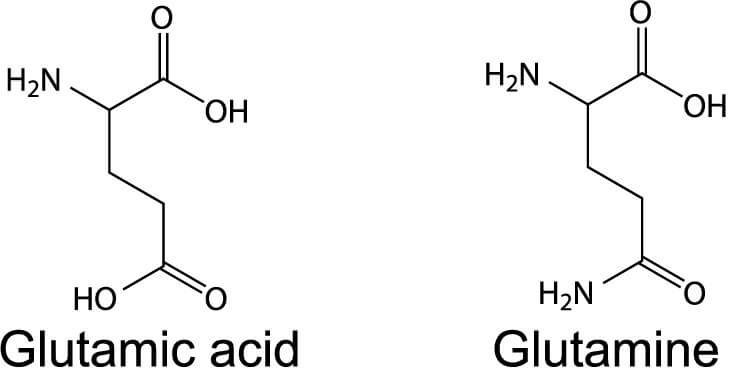
When glutamic acid loses a hydrogen ion from its carboxyl group, it forms glutamate. So, while the glutamic acid side chain has the formula CH2CH2COOH, glutamate has the formula CH2CH2COOH. In summary, glutamate is the anion of glutamic acid. The names are often used interchangeably.
Glutamic acid has a pKa value (a measure of the relative strength of an acid) of 4.1. A pKa value of 4.1 means, in environments where the pH is above 4.1, it loses its positive charge and exists mostly in its negatively charged form.
Therefore, in the human body, glutamic acid is almost always glutamate, because the conditions in the body (pH 7) favor this loss of positive charge. As a result, under physiological conditions, it is considered a polar, negatively-charged, aliphatic amino acid.
Glutamic Acid vs. Glutamine
Glutamic acid and glutamine are both amino acids that are often confused because of their similar names. Glutamine (Gln or Q) has a side chain that is similar to that of glutamic acid, except that the carboxylic acid group is replaced with an amide group (NH2).
Glutamate can be synthesized from glutamine in the central nervous system through the glutamate-glutamine cycle.
Umami Flavoring
Glutamic acid is also responsible for umami flavoring. This is the newest of the five classified flavors (the others being salty, sweet, bitter, sour). Umami is a Japanese word that loosely translates to “pleasant savory taste.” We taste this flavor in foods that are high in glutamates, such as gravies, shellfish, yeast extract, and soy sauce, through glutamate receptors. As a result, gllutamate is used as a flavor enhancer in the form of monosodium glutamate (MSG).
Biological Activity of Glutamic Acid
Glutamate as a Neurotransmitter
As well as being a building block of proteins, glutamic acid (specifically, glutamate) also acts as a neurotransmitter in the central nervous system. Glutamate is highly abundant in the brain and throughout nervous tissue, where it is the primary excitatory neurotransmitter.
Neurotransmitters are chemicals in the body that act as signaling molecules throughout the nervous tissue. Aside from glutamate, other examples of neurotransmitters include acetylcholine, adrenaline, and dopamine.
Glutamate exerts its action by binding to and activating receptors that are primarily found embedded in the cell membrane of neurons and astrocytes. These receptors fall into four classes: AMPA, kainate, NMDA, and metabotropic receptors.
As an excitatory neurotransmitter, when glutamate binds to its receptors, it increases the likelihood that the neuron will fire an action potential. Action potentials are also known as ‘nerve impulses’, and are the way that excitatory neurons pass signals to one another in order to communicate.
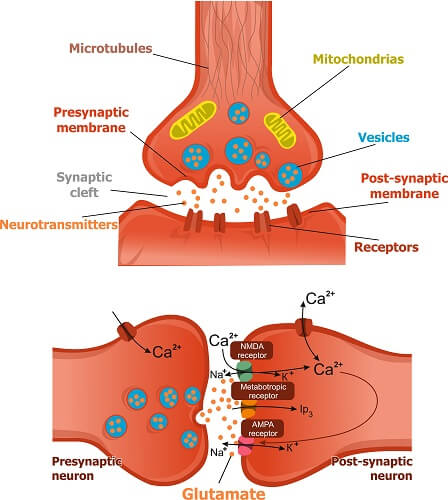
Glutamate is stored in vesicles at chemical synapses. When a nerve impulse reaches the synapse, glutamate is released into the synaptic cleft by exocytosis, where it can activate glutamate receptors in the next cell.
AMPA, kainite and NMDA receptors are ionotropic receptors, meaning that when activated, the open channels in the membrane that allow ions to pass through. Metabotropic receptors are more variable, and generally work via signaling through a second messenger.
Through its function as a neurotransmitter, glutamate is very important for synaptic plasticity, which is crucial in the processes of learning and memory.
Glutamic Acid Decarboxylase
Glutamic acid decarboxylase (also called glutamate decarboxylase) is an enzyme that catalyzes the conversion of glutamate to gamma-aminobutyric acid (GABA). Therefore, glutamate acts as a precursor for GABA – the principle inhibitory neurotransmitter in the nervous system.
GABA binds to and activates GABA receptors. Through this binding, GABA acts as an inhibitory neurotransmitter, decreasing the likelihood of a neuron firing an action potential. Increasing the activity of GABA receptors can have a sedative effect since it inhibits neuron firing. Consistently, drugs such as benzodiazepines and alcohol bind to and activate GABA receptors.
Clinical Relevance
Various disorders are associated with impaired glutamate signaling in the brain.
Neurodegenerative Disease
Glutamate is critical for most actions of the central nervous system. Chronic neuronal excitation by glutamate has been linked with degenerative disorders of the central nervous system, including motor neuron disease, Huntington’s disease, and Alzheimer’s disease. The reason for this is a phenomenon known as glutamate toxicity or excitotoxicity, which can kill nerve cells and cause chronic diseases of the nervous system.
Psychiatric Disorders
Excessive glutamate activity has also been linked to psychiatric disorders: major depressive disorder, schizophrenia, and bipolar disorder. Pharmacological compounds (for example, ketamine) that target the glutamate system are currently in clinical trials for the treatment of such psychiatric disorders.
Substance Abuse
Glutamate also has an important function in developing and maintaining addiction. This is a result of its role in cognitive processes such as reinforcement, sensitization, habit learning and reinforcement, conditioning, cravings, and relapse. Therefore, glutamatergic agents are being explored as a potential treatment to block drug-taking behaviors, reduce withdrawal symptoms, and limit relapses.
Quiz